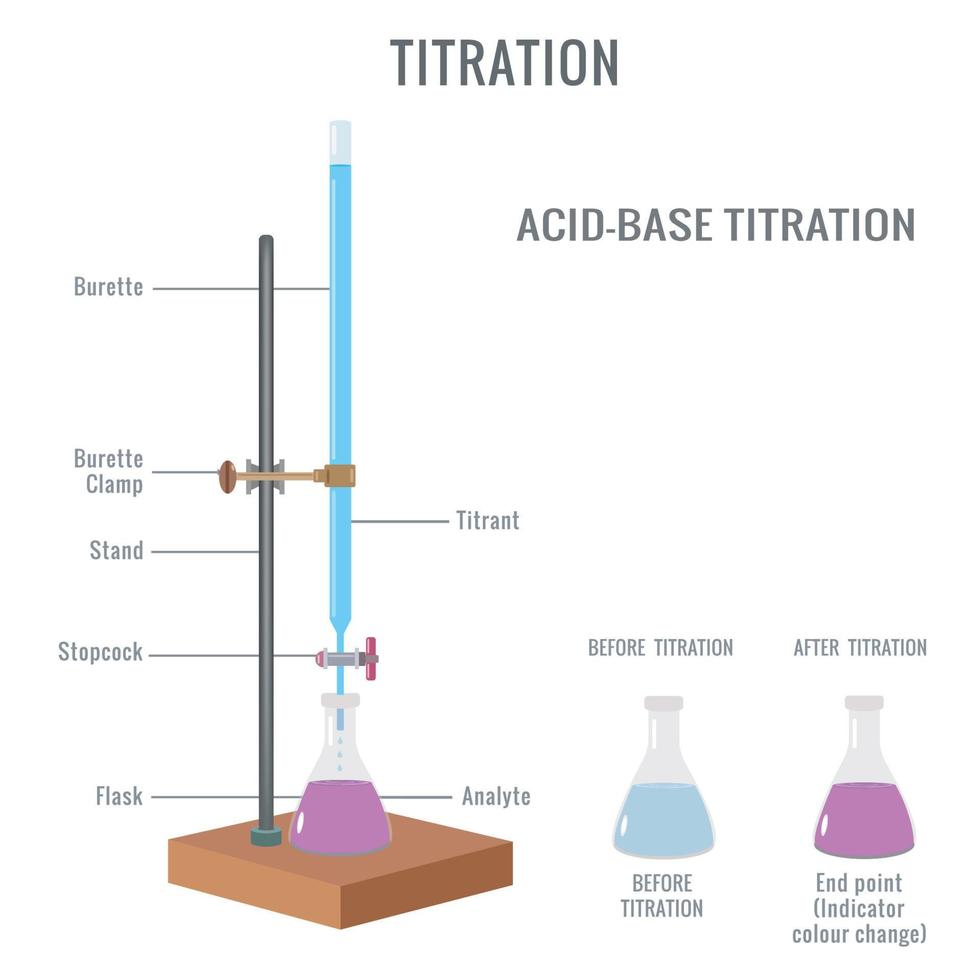
In A Titration Experiment Hcl And Lioh . Explain the function of acid. From the resulting titration curves, you will determine the. a titration is carried out for 25.00 ml of 0.100 m hcl (strong acid) with 0.100 m of a strong base naoh (the titration curve is shown in figure. Compute sample ph at important stages of a titration. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to.
from www.vecteezy.com
a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. a titration is carried out for 25.00 ml of 0.100 m hcl (strong acid) with 0.100 m of a strong base naoh (the titration curve is shown in figure. Compute sample ph at important stages of a titration. Explain the function of acid. From the resulting titration curves, you will determine the.
Acid base titration experiment and phases of color change during titration 20240684 Vector Art
In A Titration Experiment Hcl And Lioh a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in. a titration is carried out for 25.00 ml of 0.100 m hcl (strong acid) with 0.100 m of a strong base naoh (the titration curve is shown in figure. Compute sample ph at important stages of a titration. From the resulting titration curves, you will determine the. Explain the function of acid.
From exoqledql.blob.core.windows.net
Titration Base Hydrochloric Acid at Bryan Barnes blog In A Titration Experiment Hcl And Lioh Explain the function of acid. a titration is carried out for 25.00 ml of 0.100 m hcl (strong acid) with 0.100 m of a strong base naoh (the titration curve is shown in figure. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order. In A Titration Experiment Hcl And Lioh.
From about.dataclassroom.com
AcidBase Titration Lab — DataClassroom In A Titration Experiment Hcl And Lioh Compute sample ph at important stages of a titration. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in. From the resulting titration curves, you will determine the. a titration is carried out for 25.00 ml of 0.100 m hcl (strong acid) with 0.100 m. In A Titration Experiment Hcl And Lioh.
From edu.rsc.org
Titrating sodium hydroxide with hydrochloric acid Experiment RSC Education In A Titration Experiment Hcl And Lioh Compute sample ph at important stages of a titration. From the resulting titration curves, you will determine the. a titration is carried out for 25.00 ml of 0.100 m hcl (strong acid) with 0.100 m of a strong base naoh (the titration curve is shown in figure. a titration is an experiment where a volume of a solution. In A Titration Experiment Hcl And Lioh.
From edu.rsc.org
Titrating sodium hydroxide with hydrochloric acid Experiment RSC Education In A Titration Experiment Hcl And Lioh Explain the function of acid. a titration is carried out for 25.00 ml of 0.100 m hcl (strong acid) with 0.100 m of a strong base naoh (the titration curve is shown in figure. Compute sample ph at important stages of a titration. From the resulting titration curves, you will determine the. a titration is an experiment where. In A Titration Experiment Hcl And Lioh.
From www.chegg.com
Solved The titration of an acid (H2A) with LiOH solution In A Titration Experiment Hcl And Lioh Compute sample ph at important stages of a titration. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. From the resulting titration curves, you will determine the. a titration is an experiment where a volume of a solution of known concentration is. In A Titration Experiment Hcl And Lioh.
From chemistrymadesimple.net
What is Titration and How is it Done? Chemistry Made Simple In A Titration Experiment Hcl And Lioh Explain the function of acid. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in. From the resulting titration curves,. In A Titration Experiment Hcl And Lioh.
From psiberg.com
The Equivalence Point Acid/Base Titrations PSIBERG In A Titration Experiment Hcl And Lioh Explain the function of acid. Compute sample ph at important stages of a titration. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. a titration is carried out for 25.00 ml of 0.100 m hcl (strong acid) with 0.100 m of a. In A Titration Experiment Hcl And Lioh.
From www.toppr.com
The heat of neutralization of LiOH and HCl at 25^∘ C is 34.868 KJ mol^1 . The heat ionization In A Titration Experiment Hcl And Lioh a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. a titration is carried out for 25.00 ml of 0.100 m hcl (strong acid) with 0.100 m of a strong base naoh (the titration curve is shown in figure. From the resulting titration. In A Titration Experiment Hcl And Lioh.
From www.dreamstime.com
Phenolphthalein Indicator in Acidbase Titration Stock Vector Illustration of labeled, diagram In A Titration Experiment Hcl And Lioh a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in. From the resulting titration curves, you will determine the. Compute sample ph at important stages of a titration. a titration is carried out for 25.00 ml of 0.100 m hcl (strong acid) with 0.100 m. In A Titration Experiment Hcl And Lioh.
From www.leybold-shop.com
Titration of hydrochloric acid with sodium hydroxide solution Titrations Acids and bases In A Titration Experiment Hcl And Lioh Explain the function of acid. From the resulting titration curves, you will determine the. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in. Compute sample ph at important stages of a titration. a titration is an experiment where a volume of a solution of. In A Titration Experiment Hcl And Lioh.
From www.scribd.com
titration ( chemistry Experiment report) Titration Analysis In A Titration Experiment Hcl And Lioh a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. From the resulting titration curves, you will determine the. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in. Explain. In A Titration Experiment Hcl And Lioh.
From www.vecteezy.com
Acid base titration experiment and phases of color change during titration 20240684 Vector Art In A Titration Experiment Hcl And Lioh a titration is carried out for 25.00 ml of 0.100 m hcl (strong acid) with 0.100 m of a strong base naoh (the titration curve is shown in figure. Explain the function of acid. From the resulting titration curves, you will determine the. a titration is an experiment where a volume of a solution of known concentration is. In A Titration Experiment Hcl And Lioh.
From www.chemicals.co.uk
Titration Experiments In Chemistry The Chemistry Blog In A Titration Experiment Hcl And Lioh a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. From the resulting titration curves, you will determine the. Explain the function of acid. a titration is carried out for 25.00 ml of 0.100 m hcl (strong acid) with 0.100 m of a. In A Titration Experiment Hcl And Lioh.
From printablehaferbrotwp.z21.web.core.windows.net
How To Do Titrations In Chemistry In A Titration Experiment Hcl And Lioh a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in. Explain the function of acid. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. Compute sample ph at important. In A Titration Experiment Hcl And Lioh.
From www.researchgate.net
Variation of HCl and LiOH concentration under different feed volumes Download Scientific Diagram In A Titration Experiment Hcl And Lioh a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in. From the resulting titration curves, you will determine the. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. Compute. In A Titration Experiment Hcl And Lioh.
From byjus.com
The heat of neutralization of LiOH and HCL at 25^°C is 34.868 kJ mol`1(mole inverse).The heat of In A Titration Experiment Hcl And Lioh a titration is carried out for 25.00 ml of 0.100 m hcl (strong acid) with 0.100 m of a strong base naoh (the titration curve is shown in figure. Explain the function of acid. Compute sample ph at important stages of a titration. From the resulting titration curves, you will determine the. a titration is an experiment where. In A Titration Experiment Hcl And Lioh.
From www.numerade.com
SOLVED Which of the following titrations will result in a basic solution at the equivalence In A Titration Experiment Hcl And Lioh Explain the function of acid. From the resulting titration curves, you will determine the. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in. Compute sample ph at important stages of a titration. a titration is an experiment where a volume of a solution of. In A Titration Experiment Hcl And Lioh.
From www.numerade.com
SOLVED A chemist is performing a titration in order to determine the amount of lithium In A Titration Experiment Hcl And Lioh From the resulting titration curves, you will determine the. a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. Explain the function of acid. Compute sample ph at important stages of a titration. a titration is an experiment where a volume of a. In A Titration Experiment Hcl And Lioh.